From Templates to Approved GMP Documents
Follow the four-step workflow used by pharmaceutical teams and their CMO/CLO partners to collect, verify, and export compliant batch data.
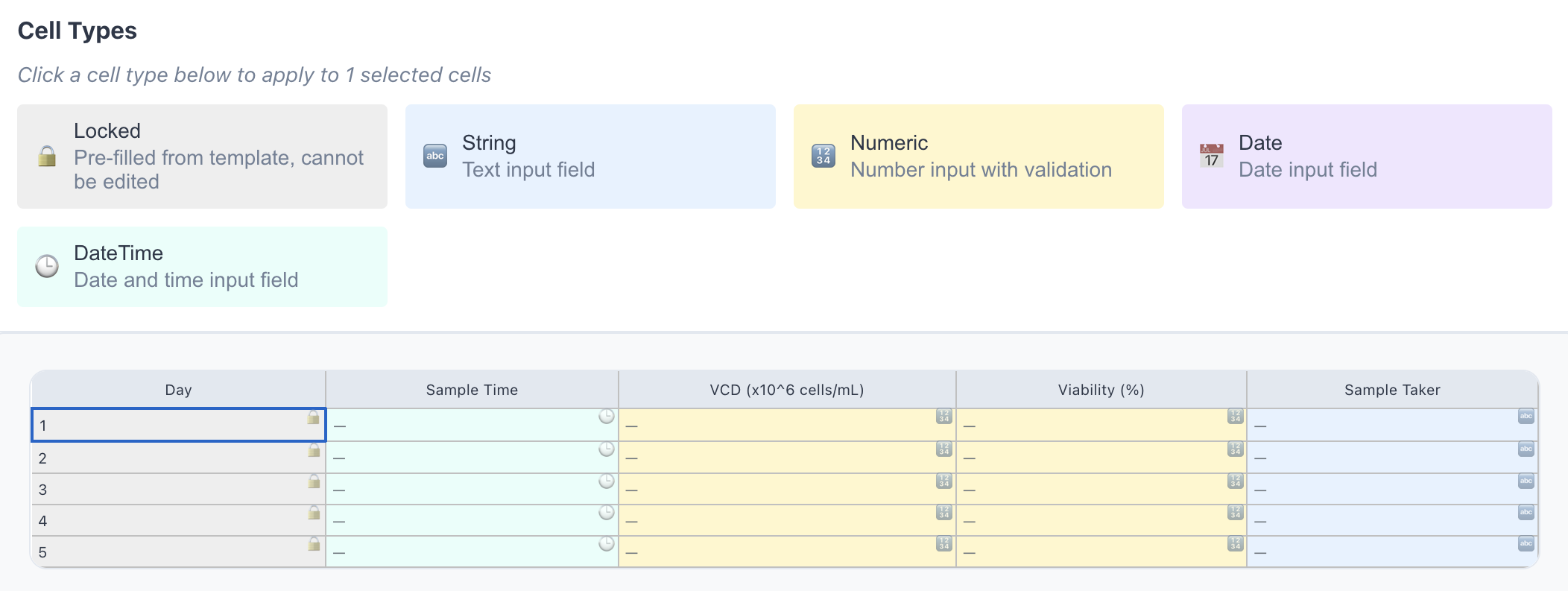
Configure Templates
Convert existing paper forms into validated digital templates in minutes — no coding required. Define field types, validation rules, and version control from a simple interface.
- Convert existing processes without coding
- Define numeric, date, text, and locked field types
- Version-controlled — changes are always traceable
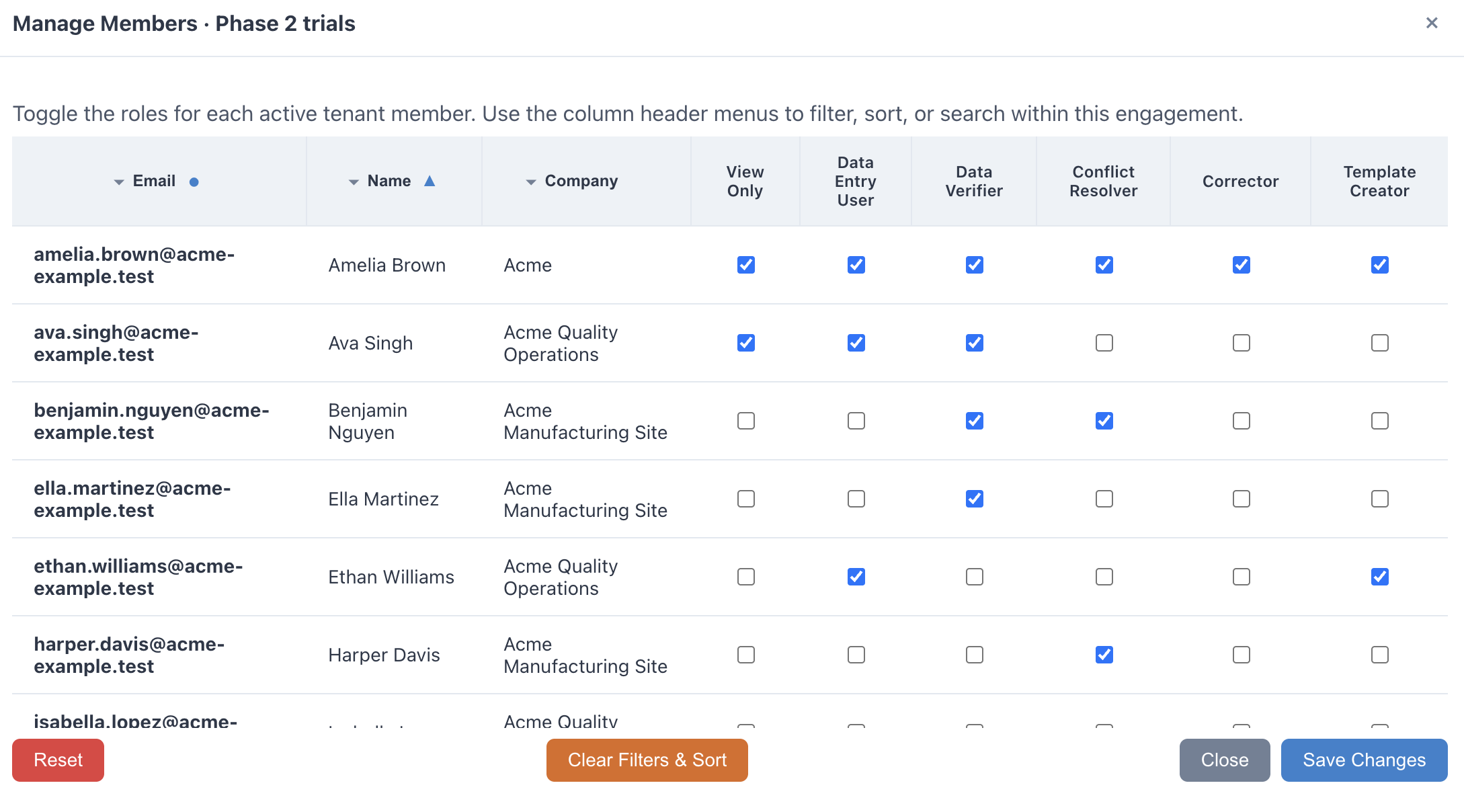
Collaborate Across Organizations
Invite your CMO and CLO partners to access templates via email. Manage role-based permissions and track submission status across every engagement in real time.
- Role-based access: Admin, Submitter, Reviewer
- Multi-organization engagements in one workspace
- Real-time visibility into submission status
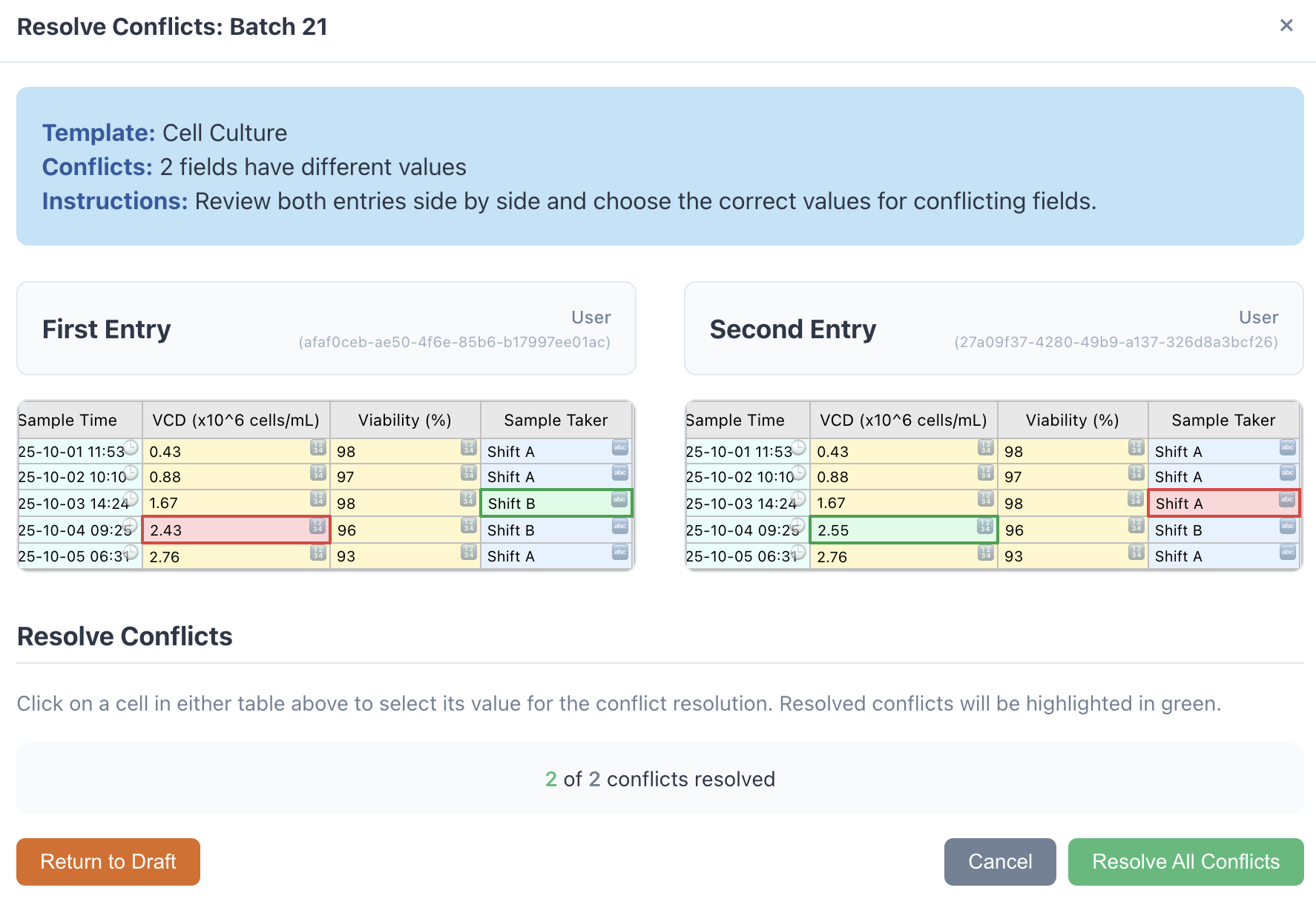
Enter and Verify Data
Collaborators fill out validated forms directly in the browser — no Excel, no paper. Double-blind entry and conflict resolution ensure every data point is independently verified.
- Real-time field validation prevents errors at entry
- Double-blind verification for critical batch records
- Third-party conflict resolution with full audit trail
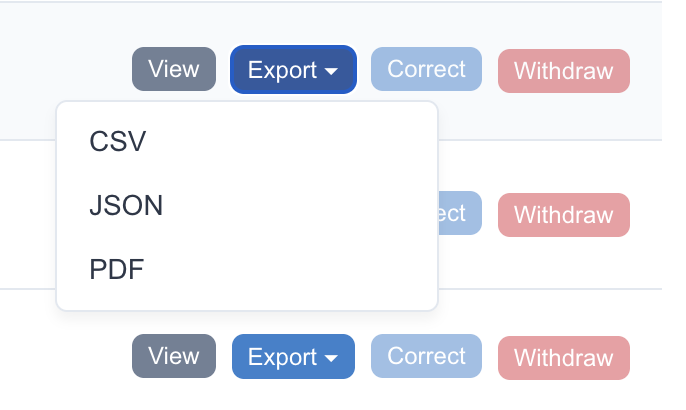
Leverage Your Data
Export verified batch records as tamper-proof PDF, CSV, or JSON — complete with audit trails and electronic signatures. Ready for regulatory filings, analytics, and audits.
- Tamper-detection hash on every PDF export
- Complete audit trail: who, what, when, and why
- 21 CFR Part 11 compliant — FDA-ready on day one
Ready to get started?
From kickoff to first live submission without a multi-month implementation.
Book a Demo Today